
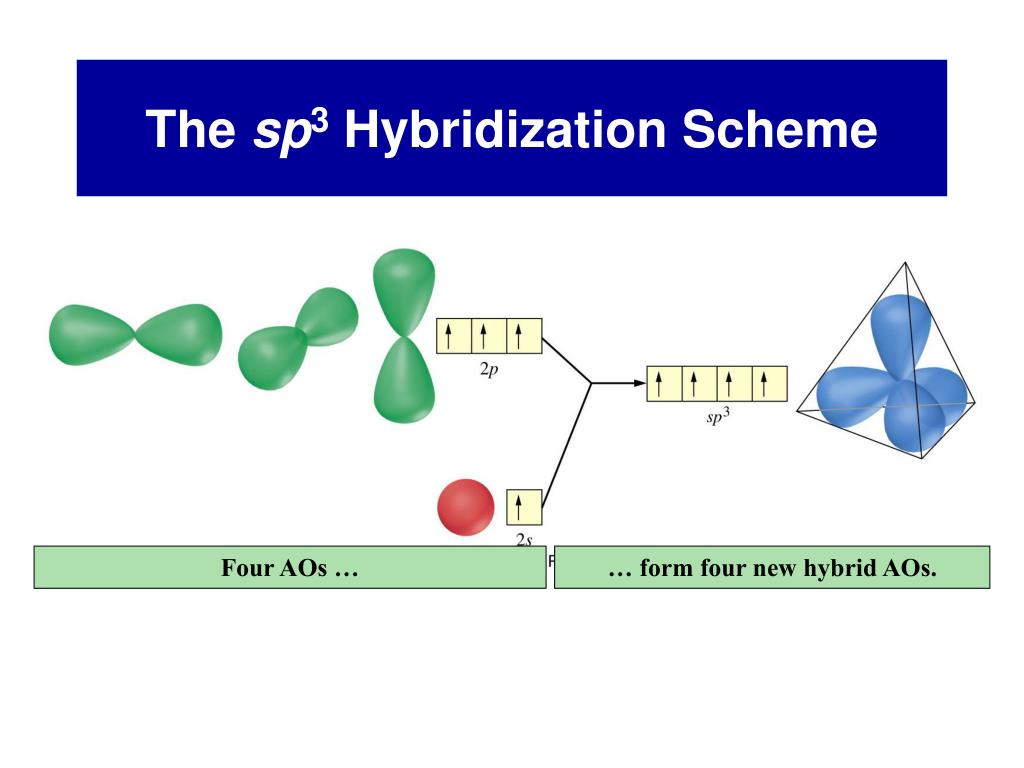
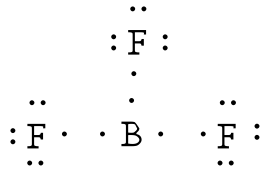
From that fact, Sulfur has the high priority Only two elements to select the center atom. Having a high valence and being the mostĮlectropositive atom are the most important facts to be the center atom. There are requirements to be the center atom in a molecule. SF 4, Total pairs of electrons are seventeen (34/2) in their valence shells. Total electron pairs are determined by dividing the number total valence electrons by two. Pairs = σ bonds + π bonds + lone pairs at valence shells valence electrons given by fluorine atoms = 7 * 4 = 28.valence electrons given by sulfur atom = 6 * 1 = 6.Is a group VIA element in the periodic table and has six electrons in its last shell (valence shell).Įlement in the periodic table and contains seven electrons in its last shell. There are only two elements in Sulfur tetrafluoride sulfur and fluorine. Total number of electrons of the valance shells of SF 4 Check the stability and minimize charges on atoms by converting lone pairs to bonds to obtain best.Mark charges on atoms if there are charges.Total electrons pairs existing as lone pairs and bonds.Find total number of electrons of the valance shells of sulfur and fluorine atom.Now, we are going to study study each step of drawing the lewis structure of SF 4. Number of steps can be changed according the complexity of the There are general guidelines to draw a lewis structure of a molecule. On atoms in SF 4 lewis structure and no overall charge in the molecule. There is a lone pair on center sulfur atom and each fluorine atom also has three lone pairs. “They opened the door to a whole new garden.In this lewis structure of SF 4, center sulfur atom has made four single bonds with four fluorine atoms. “They did not simply find a new flower or a new plant,” says Gernot Frenking of the University of Marburg in Germany, who was not involved in the work. “The material, if it could be made, would have completely different physical behaviour from an ordinary carbon chain,” Braunschweig says.Ī world of previously forbidden chemistry beckons.
BORON TETRAFLUORIDE ION BONDING ELECTRONS FREE
Next, Braunschweig and his team hope to free this boron chain from its scaffold and increase the chain length to form the boron equivalent of polyethylene, a common plastic. The secret was to attach the boron atoms to an iron scaffold, which allowed up to four to form a chain ( Nature Chemistry, DOI: 10.1038/nchem.1379). “It is extremely difficult to form chains of boron atoms,” says Braunschweig. Previous attempts to do this with boron failed and resulted in messy clusters. They also coaxed boron atoms into forming a chain. Triple-bonding is not the only way the researchers got boron to mimic its superstar neighbour, carbon, though. “It turns out that it shows a rich chemistry,” he says.
BORON TETRAFLUORIDE ION BONDING ELECTRONS FULL
In the resulting compound, each boron atom has a full suite of eight outer electrons, making it stable ( Science, DOI: 10.1126/science.1221138).īraunschweig and his team have already begun investigating the reactivity of the novel compound. Each boron atom then completed the filling of its slots by pairing up with another boron atom and pooling its original three electrons (see diagram). They filled up the empty slot by bonding each boron atom to a molecule that donated two electrons. To make triple-bonded boron that could survive at room temperature, Braunschweig and his colleagues filled all four slots. In atomic boron, one of these slots is completely empty and the other three are half-full, with one electron apiece. In 2002, Mingfei Zhou and colleagues at Fudan University in Shanghai, China, managed to make a boron triple bond – but only at 8 degrees above absolute zero.īoron can hold up to eight outer electrons: a pair in each of four slots. Boron has three outer electrons, so in principle should be capable of this too, but it has remained aloof. Carbon, with four outer electrons, and nitrogen with five, form triple covalent bonds consisting of six shared electrons, or three pairs. On the other side are the non-metals carbon and nitrogen, which prefer sharing electrons in covalent bonds.Ī single covalent bond is two electrons shared between two atoms. On one side are metals like beryllium, which give away their outermost electrons to form ionic bonds.
Until now largely obscure, boron occupies a special spot in the periodic table.
